Overcoming Corrosive Environments to Measure pH in Brine Solutions
Corrosive process environments are not new to anyone familiar with chlor-alkali manufacturing. Which produces alkali and sodium hydroxide/caustic (NaOH) by electrolysis of a salt solution. In fact, across multiple industries, manufacturers using brine must replace pH measurement devices often after corrosion and other factors have poisoned or clogged them.
But that does not need to be the case. This second article in the series dives into why corrosion has been a problem for conventional pH measurement devices — but is not a problem for innovative differential pH measurement devices.

As the brine solution undergoes electrolysis to produce chlorine, sodium hydroxide, and hydrogen, the acidity must be measured accurately for process optimization. Because pH measurements are controlling the chemical dosing. If pH measurements are inaccurate, the dosage of chemicals will be incorrect.
By successfully measuring, monitoring, and managing pH in brine solutions, manufacturers improve operation efficiency, eliminate some maintenance activities, and prevent incidents.
Corrosive Conditions Affect Conventional pH Measurement
Chlori-alkali manufacturing processes use and produce corrosive materials: hydrochloric acid (HCl) breaks down metals while chlorine (Cl2) fluid attacks plastics and elastomers. With respect to measurement devices, the conditions shorten sensor life through junction coating and plugging. This process chemistry also frequently etches the glass membrane and poisons pH sensors.
Material selection plays a significant role in the lifespan of sensor operation. Common materials in conventional pH measurement devices include Polyvinylidene Fluoride (PVDF) plastic, Teflon™ perfluoroalkoxy (PFA) industrial coatings, and general-purpose glass. Each of these materials responds to corrosion to some degree. But the real solution lies in the engineering and design of the sensor and measurement device as a whole.
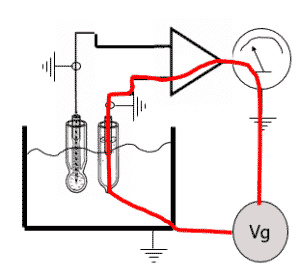
Chlorinated brine can enter the pH sensor through the porous junction, causing the Silver/Silver Chloride (Ag/AgCl) reference element’s poisoning and changing the internal Potassium Chloride (KCl) electrolyte solution. This can cause the measurement device to be sluggish or inaccurate. Double junctions, pressurized systems, and salt-bridge designs can help. But the only way to successfully combat the problem is to look for solutions with alternative reference systems.
Ground-loop currents also affect electrolysis plants. Chlorinated brine is often me asured near the electrolysis cells.Where very high currents are present that cause ground-loop currents in the pH sensor circuit, shortening the lifetime.
If the earth of the analyzer and sensor are not adequately done, multiple earth points exist — at two sensors, the analyzer, and the process pipping — in which case a potential difference creates a current that goes through and destroys the reference sensor.
Innovation Leads to the Differential pH Measurement
With the environment in mind, a recent partnership between Yokogawa and a pH measurement user has devised an innovative way to design a long-lived, accurate, consistent pH measurement device suitable for corrosive conditions.
This co-innovation led to — among other things — the removal of a traditional pH reference system while still providing stable, accurate, and reliable pH measurements. This new solution combines the measurement technology from a sodium-measuring electrode with a traditional pH measuring electrode. Also coming into play is the Yokogawa wide-body FU20 sensor’s ruggedness. The reference portion of the sensor is housed entirely in a chemically temperature-resistant glass. That provides an mV output corresponding to the process’s cation concentration.
The differential technology eliminated a junction in the process, thus eliminating the possibility of the Ag/AgCl reference becoming poisoned. Because no liquid junction is present, it cannot be coated or clogged. And there is no path for the process to affect the electrolyte.
Next in the Article Series
The ever-present high temperatures in many brine solutions should not impede the pH measurement device operation.
For helpful tips on how to extend pH sensor lifetime, download the Yokogawa pH sensor lifetime ebook for reference tips to look at.





